Human Factors Strategy
User profiles, task analysis, formative planning, and evidence-based design recommendations aligned with IEC 62366 and FDA guidance.
 Start a Project
Start a Project
Medical Device Consulting
Eleanor Williams helps medical device manufacturers reduce use risk, strengthen validation evidence, and improve readiness for Human Factors regulatory review. Support includes Class I, II, and III medical devices, with deep expertise in 510(k) and de novo pathways.
Human Factors validation activities guided through FDA approval.
Design and execution of robust programs for high-risk medical devices.
Strong cross-functional communication that drives efficient, aligned progress.
Support for the full spectrum of medical device complexity, from at home health monitoring to complex robotic surgical devices.
Core Consulting Services
User profiles, task analysis, formative planning, and evidence-based design recommendations aligned with IEC 62366 and FDA guidance.
URRA, uFMEA and ISO 14971-based risk assessment to surface, prioritize, and mitigate safety-critical use errors before submission.
Protocol design, moderation, data analysis and reporting for validation studies that stand up to regulatory scrutiny in 510(k) and de novo submissions.
Build and/or maintain ISO 13485-aligned quality management systems (QMS). Develop and/or maintain CAPA systems using robust root cause analysis to ensure effective corrective action closure.
Cutting-Edge Medical Devices
Innovation programs need Human Factors and quality execution that can keep pace with rapidly evolving technology, multi-system workflows, and global regulatory expectations.

AI Imaging + SaMD
Validation strategy for software user interfaces, alarm handling, and operator-task sequencing in algorithm-enabled imaging environments.
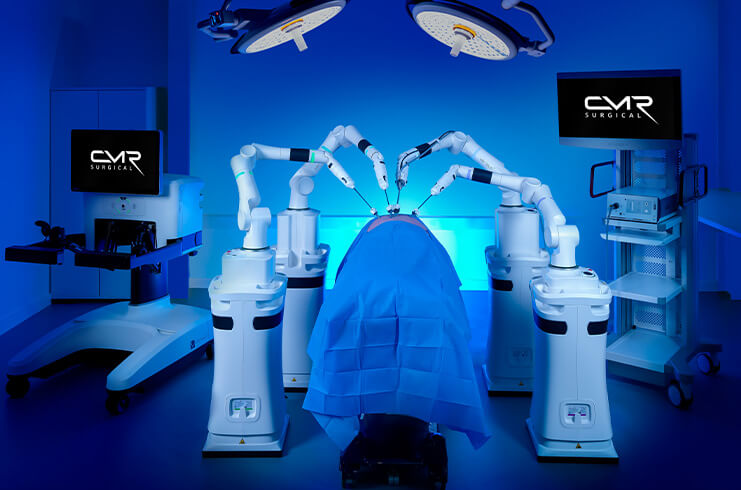
Robotic + Interventional
Formative and validation planning for teams operating across physician, technician, and nursing roles in high-acuity settings.

Connected Home Use
Human Factors evidence generation for participant device interaction, onboarding, and sustained use in real-world home environments.

Advanced IVD + Point-of-Care
Simulated-use and actual-use study execution for sample preparation, workflow reliability, and safety critical task performance.
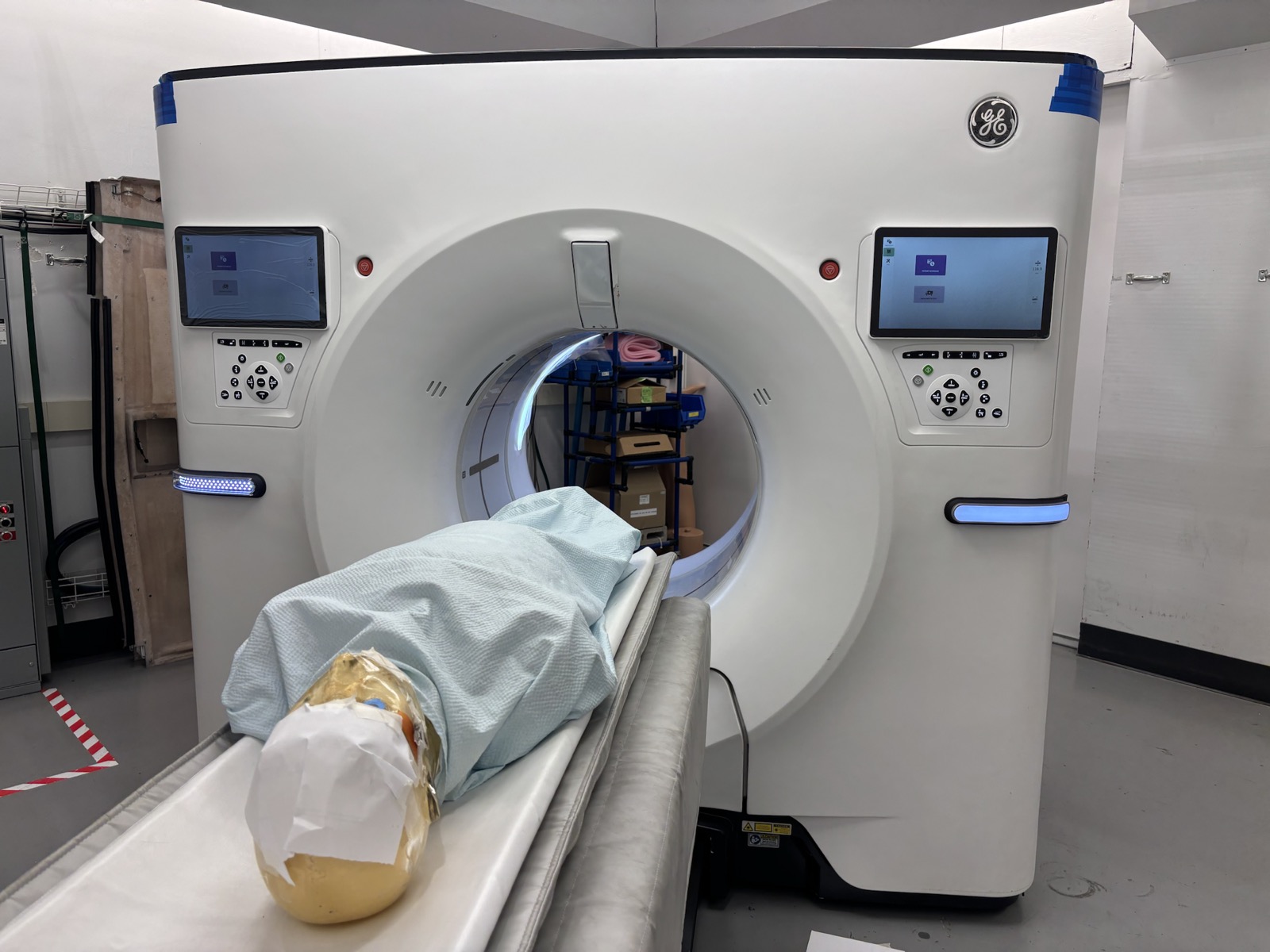
Simulated Use Environments
Simulated-use environments are configured to mirror clinical and home-use workflows, capture use-related risk signals, and generate Human Factors evidence that supports regulatory submissions.





Selected Outcomes
Led Human Factors validation activities that achieved consistent FDA approval across multiple device categories.
Rolled out Human Factors workflows for assay development and achieved full executive approval and KPI performance.
Built ISO-aligned quality frameworks from the ground up, guiding laboratory operations from 0% to 100% compliance.
Regulatory and Standards Alignment
Engagement Model
Contact
Based in Burnet, Texas. Remote and on-site consulting support for medical device manufacturers.